The New Cosmetic Regulation: What changes?
Some parts of the new Regulation have already been in force, but the major portion entered into effect on July 11, with the major aims focusing on health protection, animal protection and consumer protection.
“For the first time, the world’s largest personal care market of more than 500 million consumers will be served by a single, harmonised piece of legislation, directly applicable in all the Member States of the European Union,” comments Bertil Heerink, director-general of Cosmetics Europe, the personal care trade association.
“The regulation both gives consumers a uniform level of protection in a single market, and brings unprecedented clarity for industry.”
The new regulation should make enforcement easier and put greater confidence in cosmetics products available.
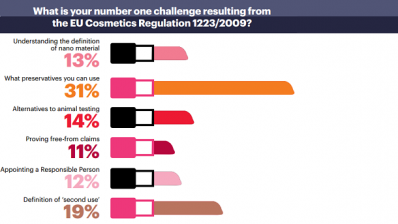
Some of the main changes will see safety requirements strengthened for cosmetic products, centralized notification of all cosmetic products placed on the EU market, the introduction of reporting of serious undesirable effects, new rules for the use of nanomaterials in cosmetic products, and the introduction of the notion of 'responsible person'.
The Commission Regulation 655/ 2013 can be viewed in full here, and the full Regulation 1223/ 2009 of the European Parliament and of the Council can be viewed here.
Please browse this gallery to take a look at some of the specific changes that come into play with the new Regulation.