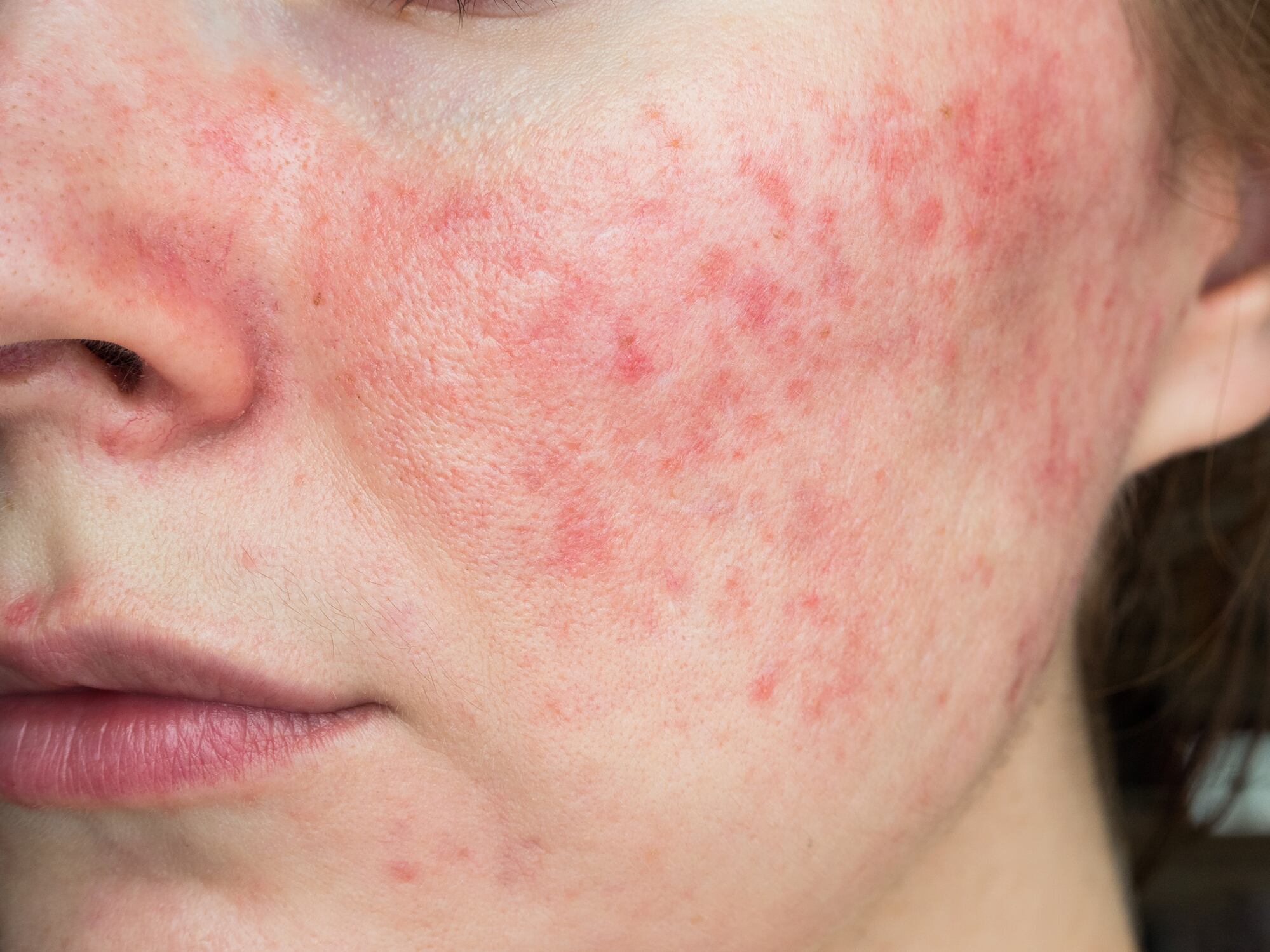
Rosacea, a skin condition that can cause flare ups, breakouts, and significant discomfort, affects the lives of over 16 million people, or 5% of the American population. In response and to support people suffering from this condition, the nonprofit organization National Rosacea Society (NRS) was established in 1992 to educate and advocate on behalf of those affected by this condition and their loved ones as well as to support medical research into better understanding and potentially finding a cure.
Last month, the NRS announced that later this year it will launch the Seal of Acceptance Program for participating cosmetic and personal care product manufacturers in support of those impacted by rosacea. The program, which offers guidance for those impacted by this condition in navigating the best fit products for rosacea sufferers, will be announced to the public soon, and applications for cosmetics and personal care product manufacturers to carry the seal on their products and materials are now open.
To learn more about the NRS Seal of Acceptance Program, including how the program was developed, how to apply to carry the seal on product packaging, and the potential impact this program can have for consumers with rosacea, CosmeticsDesign spoke with Andrew Huff, President & Executive Director of the National Rosacea Society for his insights.
Seal of Acceptance Program development
The need for the Seal of Acceptance Program was determined by demand, shared Huff. “One of the most common questions the National Rosacea Society receives from people with rosacea is what skin care and cosmetic products are appropriate to use,” he explained. To further ascertain demand, the NRS then conducted members surveys which revealed that “66% of NRS members named certain skin care or cosmetic products as triggers for rosacea signs and symptoms, and 84% said they were ‘very interested’ in more guidance on skin care.”
The new program, therefore, offers “offers a way for the NRS to provide guidance, and the early response from skin care and cosmetic companies has been very positive,” he added. Launched in accordance with the NRS’ mission to “raise awareness of rosacea, provide public health information on the disorder and support medical research that may lead to improvements in its management, prevention and potential cure,” the program is an opportunity that dovetails with the organization’s efforts to establish “April as Rosacea Awareness Month, rosacea has gone from obscurity to a condition that is routinely mentioned in health and beauty publications and on social media,” he said.
The program has been in development over the course of the last year, and the NRS has partnered with “Dr. Zoe Draelos, a well-known expert on skin care and a member of our board of directors, to develop the criteria for approval,” Huff explained. Considering that “sensitive and easily irritated skin is a common issue for people with rosacea,” there is a significant need for a process wherein products can be considered for approval or recommendation for consumer use, he said.
“Products must be free of ingredients that might damage the skin barrier or cause flushing or unwanted neurosensory stimulation such as burning or stinging,” he shared, to best alleviate any unnecessary aggravation of rosacea symptoms. Before launching the program, and “in addition to Dr. Draelos, we received advice and feedback from several leading experts in rosacea, other organizations with similar programs, and representatives from the skin care industry as we developed the Seal of Acceptance program.”
Seal Program application process
The NRS has already been in contact with a number of skin care and cosmetic companies who have expressed interest in applying for the Seal, and the program has been recently announced to the overall skin care and cosmetics industry, said Huff, with plans to announce the program to the general public later this year.
For companies interested in applying to participate in the program, they must “submit an application for each product, including a list of ingredients as presented on package labeling, the relative concentration of each ingredient in the formulation, and any ingredient safety information,” he explained. Additionally, “applicants must submit data from a human repeat insult testing (HRIPT) from at least 30 subjects as well as data from a safety-in-use test including at least 30 rosacea patients,” he said.
From there, “a panel of three independent dermatologists review each application and determine approval, and if approved, companies may license the Seal for use on product packaging and marketing materials for two years.” Additionally, “the license fee is tiered based on the company’s annual revenue,” Huff explained.
Moving forward
Ultimately, the NRS’ goal with the new Seal of Acceptance program “is to give rosacea patients — as well as the healthcare providers who treat them — an easy way to identify products that are unlikely to upset their skin,” Huff shared. Regarding skin care and cosmetic brands, program participation also offers an opportunity “to differentiate their products in the marketplace and connect with the approximately 5% of the American population who suffer from this chronic disorder.”
To learn more about the NRS Seal of Acceptance Program and for detailed guidelines for application and the approval process, visit the NRS website.