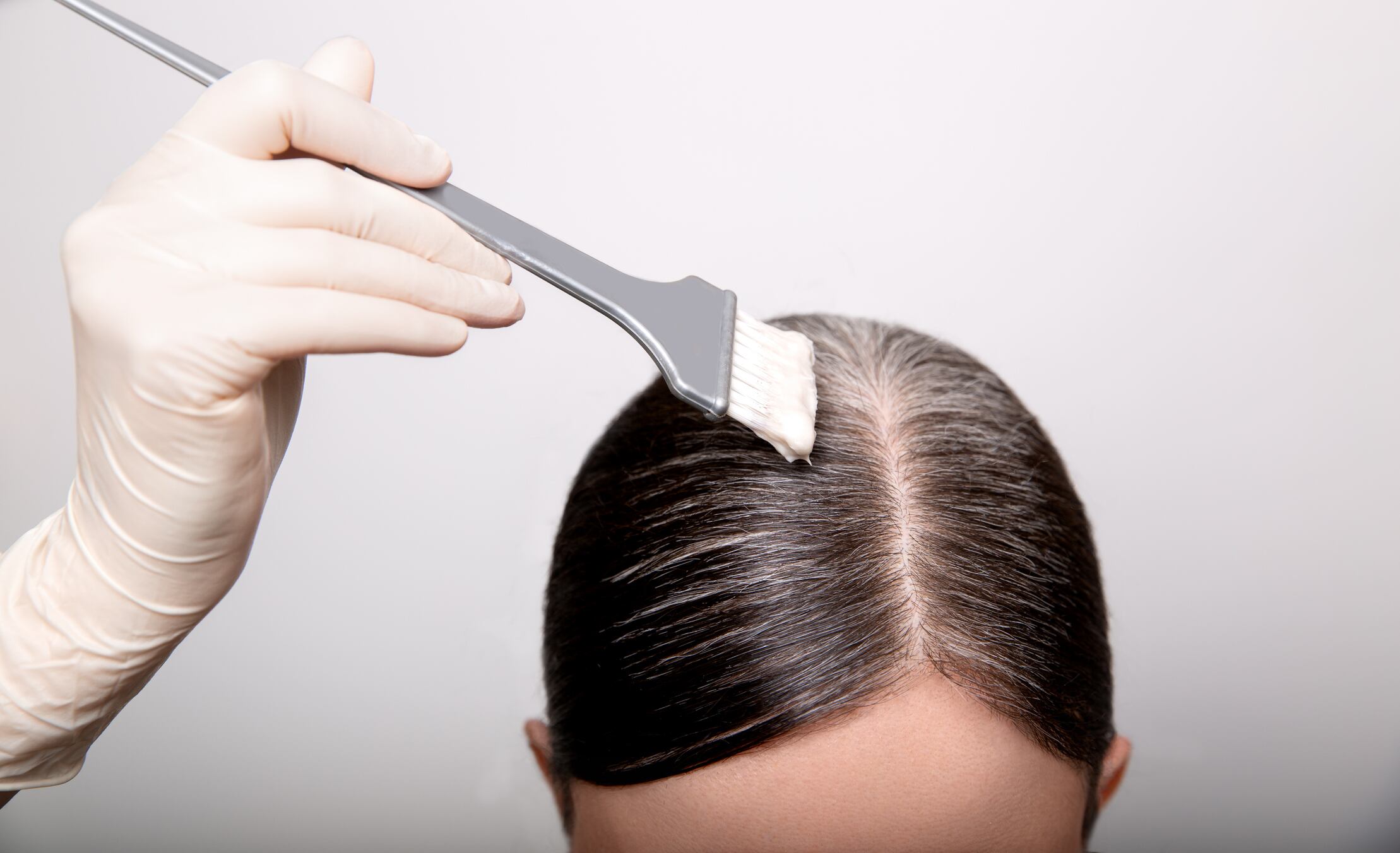
FDA originally banned lead acetate in 2018, but then stayed that ruling following objections and the request for a public hearing within the allowable timeframe. The Agency has now removed that stay and lead acetate will not be allowed for use in hair dyes effective January 6, 2022.
“We intend to exercise enforcement discretion for 12 months from the effective date to provide industry with the opportunity to deplete their current stock and reformulate their hair dye products containing lead acetate,” stated the Agency in a Constituent Update.
Lead acetate is an active ingredient that gradually darkens gray hair when used every few days. However, it can increase the level of lead in the body.
“Long overdue”
The announcement was welcomed by Melanie Benesh, a legislative attorney at the Environmental Working Group, one of the public interest groups and individuals that petitioned the Agency for the ban.
“A ban on lead acetate in off-the-shelf hair dyes is long overdue,” she said. “There is no safe level of lead exposure, which has been linked to developmental issues, reduced fertility, organ system toxicity, cancer and other serious health problems. We’re grateful for the FDA’s effort to protect public health from this source of exposure to one of the most hazardous chemicals known.”
Scott Faber, EWG’s senior vice president for government affairs, added that the federal system for regulating cosmetics safety needs reform.
“The federal law designed to ensure that personal care products are safe has remained largely unchanged since 1938,” said Faber. “It’s good news the FDA has finally banned something as dangerous as lead from a product you put on your scalp. But it’s long past time for Congress to give the FDA the power and mandate to act quickly to protect us from dangers like lead acetate.”
Faber is now urging the Agency to grant another EWG petition, this time to ban the sale of formaldehyde from hair-straightening products.
Personal Care Products Safety Act
In terms of potential reform to the regulations, the Personal Care Products Safety Act was introduced in July by Sens. Dianne Feinstein (D-Calif.) and Susan Collins (R-Maine). The act would empower the FDA to review product ingredients and provide companies with clear guidance, including whether ingredients should continue to be used and if consumer warnings are necessary. It also requires the FDA to issue recalls on products likely to cause significant harm if companies refuse to do so voluntarily – an authority the FDA currently lacks.
“We use personal care products every day, but most Americans don’t know the government lacks authority to ensure the safety of products we put on our bodies and hair,” said Senator Feinstein at the time of the introducing the legislation. “What’s particularly striking is that when the FDA finds an unsafe product, it cannot force a company to stop selling it. Our bipartisan bill will finally bring the FDA into the 21st century by giving it authority to ensure personal care products are safe.”
“By strengthening FDA oversight of the ingredients in personal care products for the first time in more than 80 years, our legislation would help protect the health of consumers, support small businesses, and provide regulatory certainty for manufacturers,” added Senator Collins.