J&J loses in court, talcum powder linked to cancer
Two products were pertinent to the case: Shower to Shower, which is now owned by Quebec-based Valeant Pharmaceuticals International (not a defendant in this case), and Johnson & Johnson’s baby powder.
Shower to Shower was a J&J brand until late 2012, when Valeant purchased it along with the Caladryl brand for $41.7m. That product is formulated with Zea mays (corn) starch, talc, sodium bicarbonate, tricalcium phosphate, fragrance, and maltodextrin, according to the Valeant product page.
Johnson & Johnson’s baby powder is formulated with only two named ingredients: talc and parfum.
Brand loyalty
Jacqueline Fox used both products for over 35 years, subsequently developed ovarian cancer, and died in October of 2015. Her son explains that she indeed used the products for years. “It just became second nature, like brushing your teeth,” he tells the press. “It's a household name.”
This is the first such case where damages were awarded. The jury awarded $72m to Fox’s family, $10m in compensatory damages and $62m in punitive damages.
At issue was the assertion that “in an effort to boost sales, [J&J] failed for decades to warn consumers that its talc-based products could cause cancer,” reports reuters.com.
The case in question, Hogans et al v. Johnson & Johnson et al, Circuit Court of the City of St. Louis, Missouri, No. 1422-CC09012, was a civil involving nearly 60 people.
Going to court
A similar case, decided in 2013, linked J&J talc products to cancer as well: “a federal jury in Sioux Falls, South Dakota found that plaintiff Deane Berg's use of Johnson & Johnson's body powder products was a factor in her developing ovarian cancer,” according to reuters.com. No damages were awarded, however.
Many other civil cases have been filed—close to 1,000 in Missouri and 200 in New Jersey. And since this week’s verdict was announced, law firms have begun soliciting complainants, “If you or somebody you know was diagnosed with ovarian cancer….”
Company line
Johnson & Johnson is expected to file an appeal. And the company has already begun a conversation about its confidence in the ingredient.
Carol Goodrich, a Johnson & Johnson spokeswoman, said after the verdict was announced: "We have no higher responsibility than the health and safety of consumers, and we are disappointed with the outcome of the trial. We sympathize with the plaintiff's family but firmly believe the safety of cosmetic talc is supported by decades of scientific evidence."
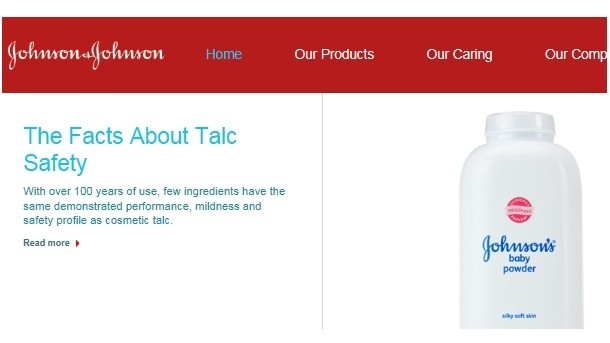
Today the J&J website prominently features a link to a blog item dated February 24, 2016 and titled, “The Facts about Talc Safety.” The post doesn’t mention the case, of course, but it lists several points strongly suggesting that J&J and the international personal care industry believe talc is a safe ingredient.
The decision in the case “goes against decades of sound science proving the safety of talc as a cosmetic ingredient in multiple products,” affirms Goodrich in her statements to the press.